In treating open skin wounds in small animals, one of the most important decisions you make is bandage selection. It’s easy to think of bandaging as a routine step after wound preparation, but in reality, the bandage plays an active role in how the wound heals. The right bandage can help debride, control infection, manage exudate, and protect developing tissue. The wrong bandage — or the right bandage used at the wrong time — can delay healing and create new problems.
Author
Saturday, March 28, 2026
Demystifying Bandage Selection for Wounds in Dogs and Cats
Monday, January 26, 2026
Inspiratory Stridor in an Older Labrador: Is It Laryngeal Paralysis?
Monday, May 19, 2025
The Punch Technique for Aural (Ear) Hematomas in Dogs and Cats: Simple, effective, and cosmetic!
Aural hematomas are characteristic in appearance.(Fig.1,2)
 |
| Fig. 1: Aural hematoma in a cat |
 |
| Fig. 2: Aural hematoma in a young white tiger |
The pinna is enlarged and fluctuant. Differential diagnoses include acute allergic response causing severe swelling of the pinna, and neoplasia of the pinna. Aspiration of the mass reveals blood or serum. Rule out underlying ear canal problems by a thorough palpation of the ear canal and otoscopic exam. Also, thoroughly examine the animal for evidence of skin disease such as allergies, seborrhea, fleas, or pyoderma.
Treatment
- Clip and prepare both sides of the pinna for aseptic surgery. Place a surgical sponge in the ear canal to prevent accumulation of blood.
- Use a skin biopsy punch (size 4-6 depending on the size of
the dog) to remove small plugs of skin and cartilage on the medial side of the pinna.(Fig. 4)

Creating punch incisions on the medial aspect of the pinna for drainage of aural hematoma. - Attempt to penetrate only the skin and 1 layer of the cartilage with the punch; however inadvertent removal a small section of both of the cartilage layers is not problematic.
- Make incisions about 0.5 – 1 cm apart and perform as many punches as necessary to drain the entire hematoma.
- Tack the skin edge of each incision with monofilament nylon, polypropylene, or Monocryl in a simple interrupted pattern.(Fig. 5) The size of suture can be 3-0 or 4-0 depending on the size of the animal. It is not necessary for the suture to penetrate full thickness through all layers of the pinna including the skin on both sides but the suture should incorporate both layers of cartilage and the skin on the medial surface.
 |
| Fig. 5: Suturing the edge of each punch incision with monofilament suture. |
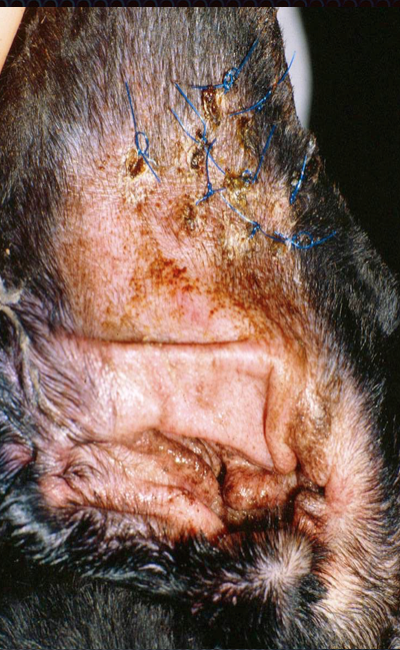 |
| Fig. 6: Pinna of a dog 2 weeks after the punch technique for aural hematoma (Photo courtesy of Dr. Daniel Smeak) |
Sunday, May 18, 2025
Scrotal Hematoma in Dogs After Castration: Why does it happen and how to prevent it.
 |
| Fig. 1: Scrotal hematoma that developed after castration in this golden retriever |
This causes excessive tissue to be incorporated in the ligatures. Insufficient stripping of the adipose tissue from the cord also predisposes the ligature to slippage.
 |
| Fig. 3: Proper exposure and clamping of the spermatic cord during routine canine castration. |
I prefer closed castration, even on large dogs, because it allows excision of the testicular tunics along with the testicle. (Fig. 5)
Friday, February 9, 2024
Introducing the Air Plasma Surgical Device (Guest Blogger Dr. William T. N. Culp)
William T. N. Culp, DVM, Diplomate, ACVS
Veterinary Teaching Hospital
UC Davis School of Veterinary Medicine
Davis, California
AirPlasma is a recently developed technique in which air is ionized to generate energy. This process is conducted without the use of gases such as argon and helium and utilizes only air as a conductor. The energy that is created is visible in the form of a glow and tissue can be vaporized with simultaneous capillary hemostasis. (Lacitignola 2020)
AirPlasma has several advantages over other forms of similar devices. The AirPlasma device works at a much lower temperature than other plasma devices, and the local thermal damage at the point of contact can be minimized with significantly less tissue necrosis. AirPlasma does not require a return plate on the patient which avoids burns on the patient’s skin encountered with some electrosurgical devices. There also is evidence that AirPlasma has a low carcinogenic potential which is particularly important as other devices can generate fumes that result in exposure to certain carcinogens. Lastly, as stated above, AirPlasma devices are able to generate energy without the need for external gases.
There are currently 3 major areas where AirPlasma is utilized in veterinary patients: ablation of tumors, cutting, dissection, and micro-coagulation. Small masses can be ablated with the device utilizing several available tip options. The device is particularly useful for cutting and dissecting tissues as the generated plasma can be highly focused on the targeted area. Because of this advantage, cutting and dissection during procedures such as total ear canal ablation, staphylectomy, and tumor removal can be very precise with minimal trauma to surrounding tissues. Finally, this device is particularly useful in assisting with microcoagulation at surgical sites after biopsies or resections. Commonly, after liver lobectomy or other partial organ resection, a small amount of bleeding can occur. Using the AirPlasma on those sites can often control minor oozing and prevent continued blood loss.
AirPlasma technology is an exciting new modality that is demonstrating promise. Proficiency is achievable in a short period of time, and safety appears to be high. Further studies will continue to elucidate outcomes with this technology, and the applications in veterinary patients are likely to increase. For more information about the AirPlasma device, and Onemytis see http://www.medviatech.com/.
Reference
Lacitignola L, Desantis S, Izzo G, et al. Comparative morphological effects of cold-blade, electrosurgical, and plasma scalpels on dog skin. Vet Sci 2020;7,8; doi:10.3390/vetsci7010008
Monday, September 4, 2023
Torsion of a Cryptorchid Testicle
Tank is a 5-month-old male intact boxer who presented to the Emergency Service for acute vomiting and abdominal pain. The vomiting began about 12 hours before presentation. The referring veterinarian had obtained abdominal radiographs and was suspicious of a gastric foreign body. There was no other history of medical problems with Tank.
On physical examination, Tank was quiet but responsive, ambulatory, and mildly dehydrated. He was painful on abdominal palpation, especially in the mid to caudal abdomen. Only one testicle was palpable in his scrotum, which was determined to be the right testicle. The left testicle was not palpable in the inguinal area.
A complete blood count and serum chemistry profile revealed no significant abnormalities. On review of the referring veterinarian's abdominal radiographs, we did not find evidence of a gastric foreign body or other abnormality.
Abdominal ultrasound revealed a structure that appeared to be a testicle. (Fig 1)
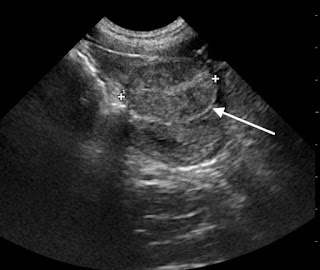 |
| Fig 1: Abdominal ultrasound showing a cryptorchid testicle (arrow) |
Placement of the ultrasound probe on the structure elicited severe pain. We were concerned that Tank had torsion of the abdominal cryptorchid testicle.
A left paramedian abdominal exploratory was performed. Torsion of the left retained testicle was confirmed, and the testicle was removed.
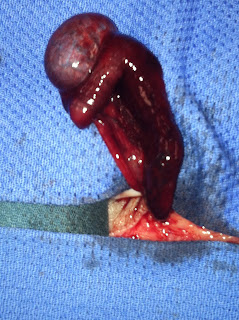 |
| Fig. 2: Surgical removal of the abdominal testicle |
The descended testicle was also removed.
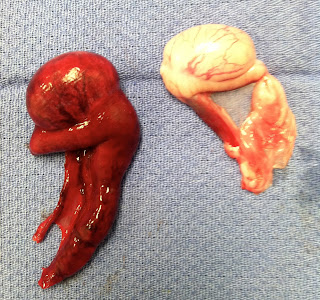 |
| Fig. 3: Both testicles removed from Tank, normal on the right, testicular torsion on the left |
Tank made an unremarkable recovery from anesthesia and surgery and was discharged from the hospital the following day.
Cryptorchidism is a common congenital anomaly in male dogs. One or both testicles can be retained in the inguinal region, inguinal canal, or abdomen. For more details on surgery for this disorder, see https://drstephenbirchard.blogspot.com/2014/07/cryptorchidism-in-dogs-5-ways-to-make.html, and scan the QR code for a complete discussion of cryptorchidism in dogs and cats.
Sunday, February 19, 2023
Adaptic Touch: The Ideal Dressing For Open Skin Wounds in Dogs and Cats
When managing open skin wounds in dogs and cats, bandaging materials and techniques are critical elements of the treatment strategy. The interior of the bandage in contact with the wound is called the primary layer and is the most important of the dressing for creating a suitable environment for healing. Many options for the primary bandage layer are available, including wet saline sponges, dry sponges, silicone dressings, medical-grade honey gauze, petroleum-impregnated gauze, and Telfa pads. The ideal first layer of the bandage should encourage granulation tissue formation. It should not inhibit healing cells and not promote infection. The primary layer should allow absorption of wound exudate into the second layer of the bandage to prevent accumulation at the wound surface.
Telfa pads have historically been used for open wounds in animals, but newer materials that allow better absorption of exudate from the wound are available. Wet saline sponges are commonly used as the initial primary bandage layer in acute traumatic wounds because they are highly absorptive, allow gentle debridement of dead tissue, and are inexpensive. However, when saline sponges become dry, they are adherent, making bandage changes painful. Patient sedation is frequently necessary to prevent discomfort during removal. Lavage of the dried sponges with sterile saline during bandage change will allow them to be peeled off the wound more easily and with less pain. Although wet saline sponges are very effective in initially managing severe wounds, such as bite wounds in dogs and cats, a primary bandage layer is needed for the next healing phase. Ideally, this material should allow absorption of wound exudate and be non-adherent to reduce patient discomfort.
 |
| Vitalitymedical.com |
The Adaptic Touch is an excellent choice for the primary bandage layer on open wounds in dogs and cats. It is a soft and pliable silicone-coated mesh material that readily conforms to the wound surface. The pore size in the mesh is small enough to prevent the ingrowth of granulation tissue into the material. The Adaptic Touch mesh allows the passage of wound exudate into the secondary bandage layer and is non-adherent, minimizing patient discomfort during bandage changes. It has been used extensively for various human wounds and is gaining favor with veterinarians. Cotton is a good choice for the secondary layer of the Adaptic Touch bandage allowing for the transmission of fluid through the pores of the mesh. Use only one layer of the Adaptic to ensure adequate absorption into the cotton.
The original Adaptic mesh is a petroleum-impregnated gauze that allows absorption and is non-adherent. However, petroleum may have some detrimental effects on wound healing, and it is only partially non-adherent. These characteristics make them less ideal than their counterpart, the Adaptic Touch.
See the above video for a demonstration of the non-adherent nature of the Adaptic Touch dressing.
In summary, the ideal bandages for open skin wounds in dogs and cats have primary layers that allow absorption of wound exudate, are non-adherent, and do not inhibit wound healing. For wounds that are highly contaminated and filled with dirt and debris that cannot be entirely removed by surgical debridement and flushing, moist saline sponges are indicated as the initial bandage. Since these bandages are adherent and painful to remove, transition to non-adherent dressings such as the Adaptic Touch is recommended for the next phase of wound treatment.
References
International case series: Using ADAPTIC TOUCH® Non-Adhering Silicone Dressing: Case Studies. London: Wounds International, 2013.
Wounds International case studies evaluation. ADAPTIC TOUCHTM Non-Adhering Silicone Dressing in skin tear management. Wounds International, 2018 (Suppl). Cooper
Janice Bianchi*, Simon Barrett, Fania Pagnamenta, Fiona Russell, Sandra Stringfellow, Pam
Cooper. Consensus guidance for the use of Adaptic Touch non-adherent dressing. Wounds UK, 2011, Vol 7, No 3
When surrounded with love, seriously ill pets are more likely to survive. Read about this in Dr. Birchard's new book. Click here for more information.





















